FDA continues to work with its partner in vaccine safety surveillance, the CDC, to monitor reports of GBS following vaccination with the Janssen COVID19 Vaccine Some COVID19 patients stricken by GuillainBarre syndrome AstraZeneca vaccine and GuillainBarré syndrome There's minimal cause for concern Recommended for youFDA did not identify an increased risk for GBS in its evaluation of the clinical trials data regarding Shingrix, which FDA evaluated prior to the vaccine's approval in 17
Fda Adds Guillain Barre Warning To J J Vaccine What To Know Time
Guillain barre syndrome and covid vaccine fda
Guillain barre syndrome and covid vaccine fda- "Reports of adverse events following use of the Janssen COVID19 Vaccine under emergency use authorization suggest an increased risk of GuillainBarré syndrome during the 42 days following FDA To Attach Warning for GuillainBarre Syndrome to Johnson & Johnson COVID19 Vaccine The risk for developing the rare neurological condition are low following vaccination, with the FDA noting the benefits of vaccination far outweigh the potential risks Officials with the FDA are planning to attach a warning for GuillainBarre syndrome to the Johnson & Johnson




Fda Adds Warning About Rare Reaction To J J Covid 19 Vaccine Wgn Tv
The Food and Drug Administration warned on Monday that Johnson & Johnson's coronavirus vaccine can lead to an increased risk of a rare neurological condition known as GuillainBarré syndrome FDA has added a warning to Johnson & Johnson's Covid19 vaccine fact sheet after receiving preliminary reports of patients developing the rare neurological condition GuillainBarré syndrome after Regulators say the benefits of the vaccine still outweigh any risks GuillainBarré syndrome (GBS) is a rare neurological disorder in which the immune system attacks the nerves It can cause muscle weakness and sometimes paralysis People usually recover from it, but it can lead to hospitalization and, sometimes, permanent damage to nerve cells
Editor's note Find the latest COVID19 news and guidance in Medscape's Coronavirus Resource Center Although GuillainBarré syndrome (GBS) may rarely follow a recent infection with SARSCoV2, aOn , the FDA announced revisions to the vaccine recipient and vaccination provider fact sheets for the Johnson & Johnson (Janssen) COVID19 Vaccine to include information pertaining to an observed increased risk of GuillainBarré syndrome following vaccination 1 GuillainBarré syndrome is a rare disorder where the body's immune system damages nerve FDA Warns J&J Covid19 Vaccine Raises Risk of GuillainBarré Syndrome Complication The rare neurological condition is a disorder in which the
Also read FDA alerts possible link of GuillainBarré syndrome with Johnson & Johnson vaccine All of them were over 50 years old and frequently experienced facial paralysis and had a demyelinating form of the disease At the time of hospital admission, 73% of GBS patients with a Covid19 infection had increased inflammatory markers The CDC says the agency has received about 100 reports of GuillainBarre syndrome among 128 million doses of the Johnson & JohnsonJanssen COVID19 vaccineA Covid19 infection may prompt autoimmune disorder, the GuillainBarre syndrome (GBS), finds a studyIn the disorder, a person's immune system attacks the



Fda Warns Of Guillain Barre Risk For Johnson Johnson Vaccine




New Covid 19 Vaccine Warnings Don T Mean It S Unsafe They Mean The System To Report Side Effects Is Working
The FDA has released a new warning that the onedose Johnson & Johnson COVID19 vaccine could increase people's risk of developing GuillainBarré syndrome, a Meanwhile, COVID19 vaccines have been associated with GuillainBarré Syndrome The European Medicines Agency (EMA) has classified it as an "extremely unusual" adverse effect of the AstraZeneca In December , the FDA approved two vaccines for the prevention of COVID19 infection In the clinical trials of the vaccine, multiple side effects have been reported ranging from mild symptoms including but not limited to injection site pain, myalgia, fatigue, and fever to more serious side effects including anaphylactic shock 4,5 However, GuillainBarre Syndrome



What Are The Symptoms Of Guillain Barre Syndrome Should I Still Get The J J Vaccine Marketwatch



Fda Adds Warning Of Rare Neurological Side Effect To J J Covid Jab
Other vaccines have been associated with GuillainBarré syndrome Although the Johnson & Johnson shot is the only COVID19 vaccine that has been linked to GuillainBarré syndrome, it's not the Officials at the US Food and Drug Administration warned of a rare but serious neurological disorder linked to Johnson & Johnson's coronavirus vaccineWe report the first case of Guillain–Barre Syndrome after receiving the second dose of the Pfizer COVID19 vaccine, in a 42yearold woman presenting with




Fda Expected To Issue New Warning On J J Covid Jab Business And Economy News Al Jazeera



J J Covid Vaccine Raises Risk Of Rare Neurological Disorder Warns Fda The Hindu Businessline
Guillain barre syndrome and covid vaccine uk Dear Editor, GuillainBarré syndrome (GBS) is the most common and severe acute paralytic neuropathy, with about 100,000 people developing the disorder every year It is intended to tell you to pay attention to your body whether you get the vaccine, get COVID, or do nothing Because the scary fact is Covid could be an occasional trigger of GuillainBarré syndrome (GBS), a study has claimed The autoimmune disease, which can leave patients paralysed and in crippling pain, has also been linked




Fda Adds Warning About Rare Reaction To J J Covid 19 Vaccine Wgn Tv




Covid Vaccine Updates Fda Warns Of Rare Guillain Barre Syndrome Reaction To Johnson Johnson Coronavirus Vaccine Abc7 New York




What Is Guillain Barre Syndrome And Its Symptoms Miami Herald



Fda Labels Johnson Johnson Vaccine With New Rare Side Effect



Cdc Fda Looking Into Risk Of Nerve Complication After Johnson Johnson Covid 19 Vaccine




J J Covid Vaccine Fda To Announce New Warning Related To A Rare Autoimmune Disorder Report Says




Fda Will Attach Warning Of Rare Nerve Syndrome To Johnson Johnson S Covid 19 Vaccine Baltimore Sun



Us Warns Of Link Between J J Vaccine And Guillain Barre Syndrome World English Edition Agencia Efe



Fda Warning About J J Vaccine Creates More Challenges For Fighting Covid Crisis




Fda Warns Of An Increased Risk Of Guillain Barre Syndrome Following Vaccination With The J J Vaccine Healthcare Finance News




Us Fda Adds Warning On Johnson Johnson Vaccine Related To Rare Neurological Disorder Guillain Barre Syndrome The Economic Times




The Link Between The Johnson Johnson Vaccine And Guillain Barre Syndrome Shape



Fda Adds Warning Of Rare Reaction Risk To J J Covid 19 Vaccine The Hindu



New Covid 19 Vaccine Warnings Don T Mean It S Unsafe They Mean The System To Report Side Effects Is Working



Rare Cases Of Guillain Barre Syndrome Reported In Janssen Covid 19 Vaccine




Foundation Hosted Patient Listening Session On Guillain Barre Syndrome With The Fda Gbs Cidp Foundation International




Fda Adds Guillain Barre Warning To J J Vaccine What To Know Time




Fda Adds Guillain Barre Syndrome Warning To J J Covid Shot Politico




What To Know About Guillain Barre And The Covid Vaccine Cleveland Clinic




J J Covid Vaccine Warning Fda Adds Info On Rare Reaction Risk 9news Com



Fda To Warn J J Vaccine Can Increase Guillain Barre Risk Media




Fda Adds J J Covid 19 Vaccine Warning Cidrap




Covid 19 Vaccine Safety Now Soft Rollout Not Pause After Reports Of Guillain Barre Syndrome Pink Sheet




Fda Adds Warning About Rare Reaction To J J Covid 19 Vaccine Wpri Com



Johnson Johnson Covid Vaccine And Guillain Barre Everything To Know




Fda To Warn J J Vaccine Can Lead To Increased Risk Of Rare Syndrome



Chart Covid Vaccines Compared




Fda Set To Add Guillain Barre Warning To J J Covid Vaccine Health News Us News




Janssen Vaccine Increases Risk Of Guillain Barre Syndrome



Fda Lists Rare Neurological Condition As Potential Side Effect Of J J S Covid 19 Vaccine Pmlive




J J Covid 19 Vaccine May Trigger Neurological Condition In Rare Cases



Fda Warns Of Possible Rare Association Between Johnson Johnson Vaccine And Guillain Barre Syndrome




Johnson Johnson Vaccine Increases Risk Of Rare Nerve Disorder Us Fda Warns




Fda Links J J Vaccine To Rare Cases Of Guillain Barre Drug Discovery And Development




Fda Warns About J J Covid Vaccine And Guillain Barre Syndrome Risk Health Com




Fda Adds Warning Of Rare Diagnosis Of Guillain Barre Syndrome To Johnson Johnson Covid 19 Vaccine Youtube



J J Vaccine Side Effects Guillain Barre Syndrome And More Nbc Chicago




J J Covid 19 Vaccine May Trigger Neurological Condition In Rare Cases




Stakeholder Call Guillain Barre Syndrome Gbs Covid 19 Vaccine Updates 7 15 21 Youtube




Pharmacy Times Fda To Attach Warning For Guillain Barre Syndrome To Johnson Johnson Covid 19 Vaccine Learn More T Co 8dxxouuzyf T Co Pkodk1dhbd



Guillain Barre Syndrome Fda Warns Of Potential Rare Neurological Complication With Johnson Johnson Coronavirus Vaccine Cnn




Neuropsychiatrist On Incredibly Rare Condition Linked To Johnson Johnson Covid 19 Vaccine Cbs News




U S Adds Warning To J J Covid 19 Vaccine Over Small Possible Risk Of Guillain Barre Syndrome Ktla




Guillain Barre Syndrome Linked To Johnson Johnson Covid Vaccine




Learn More About Covid 19 Vaccines From The Fda Fda




Coronavirus Johnson Johnson S Covid Shot Gets Fda Warning About Rare Immune Disorder



J J Covid Shot To Carry Guillain Barre Warning Chemanager



Guillain Barre Syndrome Johnson And Johnson Vaccine Fit Increase Risk Of Nerve Disorder Us Fda Warn c News Pidgin




Johnson Johnson S Covid 19 Vaccine Has Another Problematic Side Effect
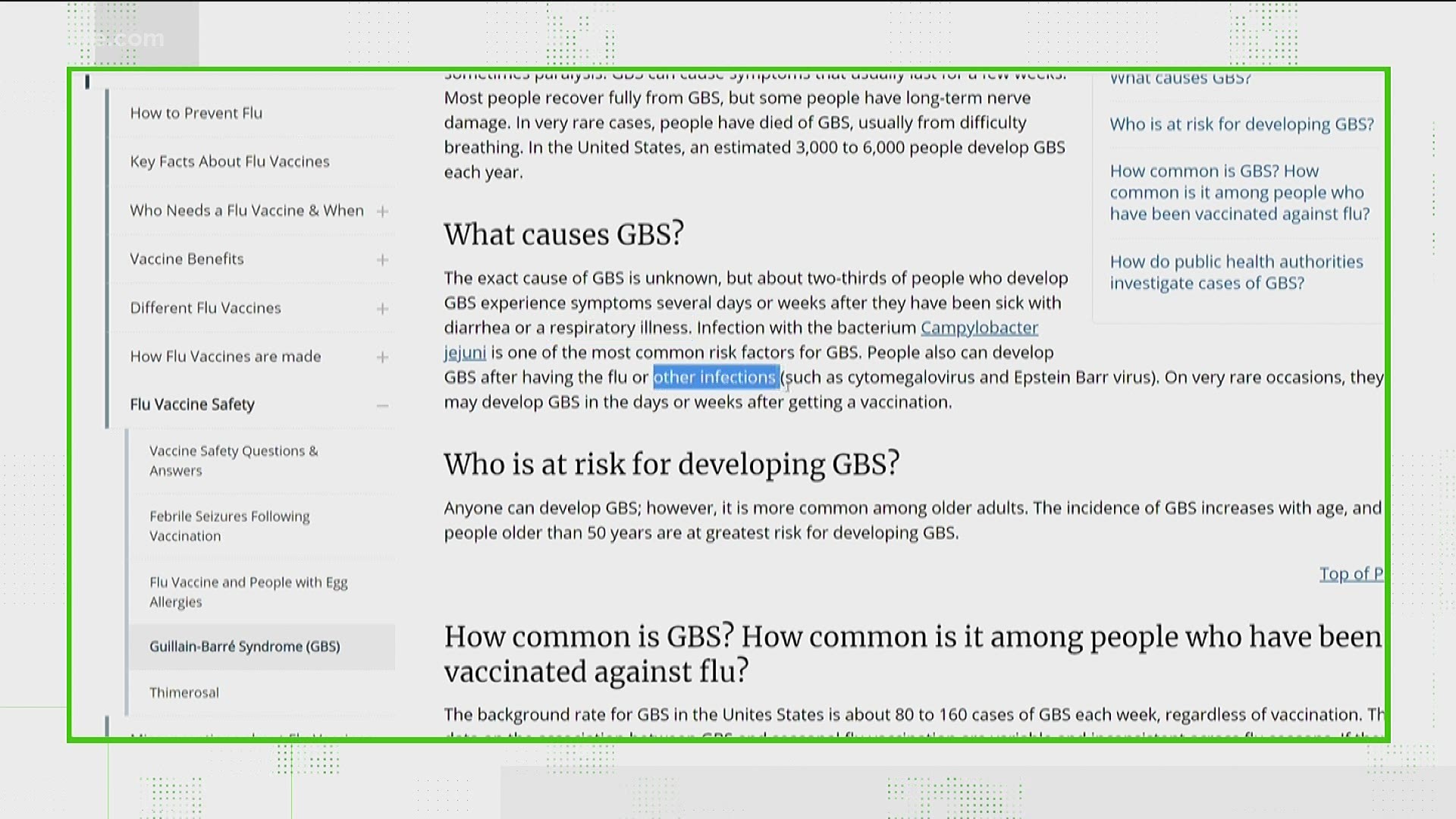



J J Warning And The Guillain Barre Syndrome 11alive Com




Fda Adds Warning About Rare Reaction To J J Covid 19 Vaccine




Fda Issues New Fact Sheet Due To Increase Risk Of Guillain Barre Syndrome With Janssen Covid 19 Vaccine Williamsburg Yorktown Daily



Guillain Barre Syndrome A Sudden Paralysis Of The Arms And Legs Science In Depth Reporting On Science And Technology Dw 13 07 21




Guillain Barre Syndrome And The Johnson Johnson Vaccine What To Know The New York Times




F D A Attaches Warning Of Rare Nerve Syndrome To Johnson Johnson Covid Vaccine The New York Times




Johnson Johnson Covid 19 Vaccine Holds The Risk Of Paralysis Fda Articles



Fda Adds Warning To J J Covid 19 Vaccine After Gbs Cases Cgtn



J J Vaccine And Guillain Barre Syndrome Information On The Fda Warning News Yale Medicine



Fda Adds Warning About Rare Reaction To J J Covid 19 Vaccine




Fda Johnson Johnson Covid 19 Vaccine Tied To Guillain Barre Syndrome




Johnson Johnson In Discussion With Fda Regarding Covid 19 Vaccine Side Effects Reuters




The Fda Warns That Johnson And Johnson Covid Vaccine May Be Linked To Guillain Barre Syndrome Wusa9 Com



J J Covid Vaccine Warning Fda Adds Info On Rare Reaction Risk Wcnc Com



Fda To Add Warning Of Guillain Barre Syndrome To J J Vaccine




Fda Issues Guillain Barre Warning On J J Covid 19 Vaccine




Fda Warns J J Covid 19 Vaccine Raises Risk Of Guillain Barre Syndrome Complication Wsj




Fda Update Warns Of Potential Rare Nerve Side Effect For J J Covid Vaccine Medcity News




J J Covid Vaccine Warning Fda Adds Info On Rare Reaction Risk Fox61 Com




What Is Guillain Barre Syndrome Cbs19 Tv




J J Covid Vaccine May Pose Small Possible Risk Of Rare Neurological Syndrome Cdc Says Cbc News




Explained What Is Guillain Barre Syndrome Why It S Being Linked To J J Vaccine



The Johnson Johnson Vaccine And Guillain Barre Syndrome What Michigan Needs To Know Bridge Michigan



Fda Will Announce Rare Incidence Of Guillain Barre Linked To Johnson Johnson Vaccine




Fda Adds Warning Of Rare Reaction Risk To J J Covid Vaccine




Johnson Johnson Vaccine Increased Risk Of Developing Guillain Barre Syndrome The Fda Says Sortiraparis Com



Us Warns Of Link Between J J Vaccine And Guillain Barre Syndrome World English Edition Agencia Efe



Johnson Johnson Covid 19 Vaccine May Lead To Guillain Barre Syndrome Fda To Issue Warning



Fda Warns About J J Covid Vaccine And Guillain Barre Syndrome Risk Health Com




What Is Guillain Barre Syndrome The Rare Condition That The Fda Have Linked To The Johnson Johnson Vaccine As Com




Fda Adds Warning Of Rare Neurological Reaction To J J Covid 19 Vaccine Business Standard News



Fda Updates J J Covid 19 Vaccine Label To Include Rare Disorder



Fda Adds Warning Of Rare Reaction Risk To J J Covid Vaccine World News India Tv




Fda Warns About Post Covid Vax Guillain Barre Syndrome Medpage Today




Had The J J Vaccine What You Should Know About Latest Risk



Fda Adds Guillain Barre Syndrome Warning To J J Covid Vaccine Axios




Case Reports Describe Unusual Guillain Barre Variants Following Covid 19 Vaccination




R I Approves Moderna Covid 19 Vaccine For Teens Ahead Of Fda Authorization The Boston Globe




Fda Adds New Warning Of Rare Nerve Syndrome To Johnson Johnson Covid 19 Vaccine




Cdc Fda Looking Into Risk Of Rare Nerve Complication After Johnson Johnson Covid 19 Vaccine



In Response To Rare Guillain Barre Cases Fda Plans New Warning For J J Covid 19 Vaccine Biospace




Vaccines Are Just As Effective Against The Delta Variant As Long As It S A Full Dose Poynter



What Do You Need To Know About The Guillain Barre Disease And Johnson And Johnson Vaccines Technology News Firstpost
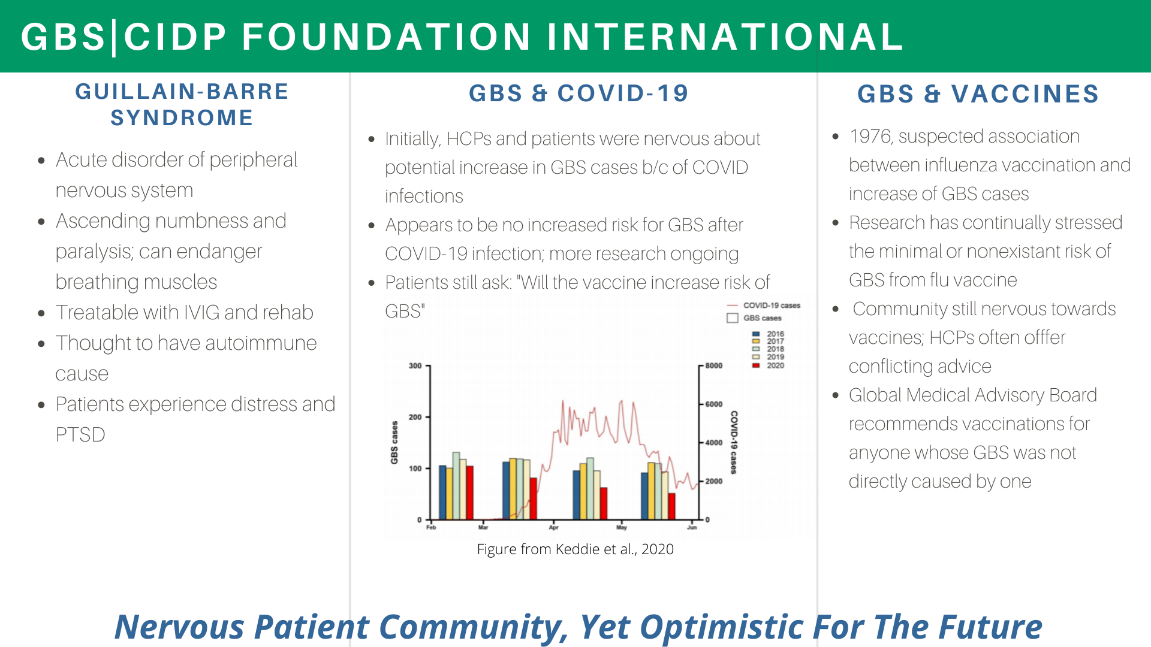



Gbs Cidp Foundation Staff Patients Collaborate With Fda Gbs Cidp Foundation International
:no_upscale()/cdn.vox-cdn.com/uploads/chorus_image/image/69574976/1232865115.0.jpg)



Fda Adds Warning About A Nerve Condition To The Johnson Johnson Covid 19 Vaccine The Verge



0 件のコメント:
コメントを投稿